AUDENZ is an intramuscularly administered, non-active, single-dose, form of influenza vaccine produced using Madin Darby Canine Kidney (MDCK) cells, a continual cell line. The cells had been modified to cultivate uninhibitedly in the culture medium while hanging in the air. The ß-propiolactone deactivates the virus, cetyltrimethylammonium bromide destroys it, and the purification is accomplished with a series of steps.
The influenza agent in AUDENZ is created through a similar manufacturing method as FLUCELVAX® and FLUCELVAX® QUADRIVALENT, which are sanitized seasonal influenza vaccines approved for use in the United States.
Indications
AUDENZ™ is a vaccine that is used to receive active immunization against flu caused by the influenza A virus H5N1 subtype. AUDENZ has been authorized for individuals over 6 months of age who are at greater danger of being exposed to the influenza A H5N1 virus strain which is included in the shot.
It is acceptable to administer AUDENZ to people aged 6 months to 17 years old, due to the immunological reactions it produces, as shown in clinical tests. The efficacy of the seasonal vaccine created following the same methodology has not been corroborated for this population. This age group’s use of the product must be confirmed by additional trials in order to be granted extended approval. The trials must demonstrate the product’s positive effects.
Dosage and Administration
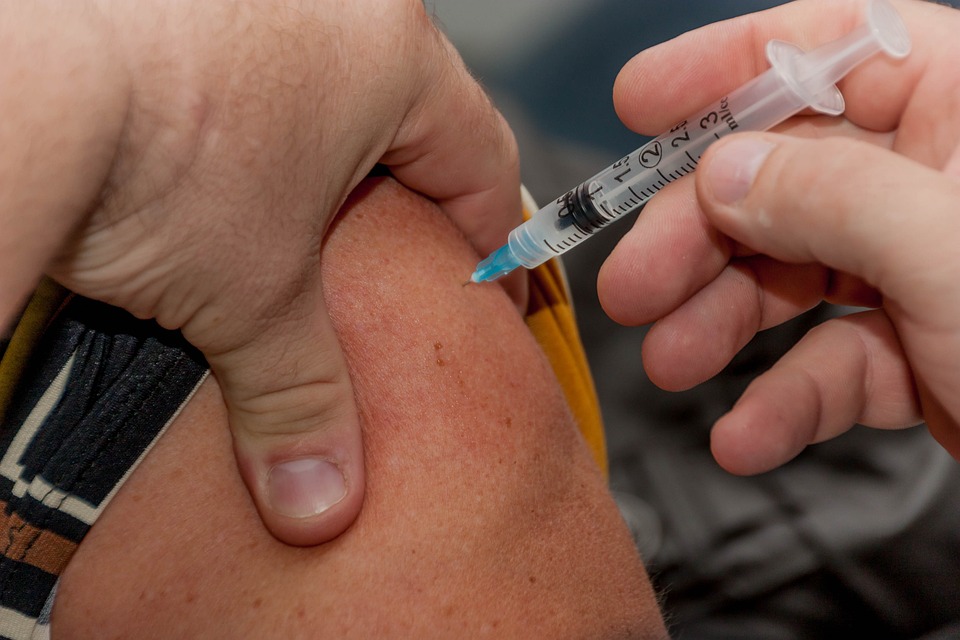
For intramuscular injection only
Give two injections of AUDENZ, 0.5 mL each, spaced 21 days apart.
Gently shake the syringe. AUDENZ has a milky-white appearance. Before administering parenteral drug products, it is important to take a look at it and check for any particulate matter and discoloration if possible (please refer to the corresponding description). If either condition exists, AUDENZ should not be administered.
The vaccine should be administered by intramuscular injection. Do not administer the vaccine into the buttocks or any area close to important nerves. For those above one year of age, the best location for an injection is the deltoid muscle at the upper part of the arm. For those aged between 6 months and 11 months, the most suitable site for an injection is the anterolateral area of the thigh.
Side Effects
Clinical Trials Experience
There is no single standard for clinical trials, so it is not possible to accurately compare adverse reactions from one vaccine to another. These results do not necessarily accurately reflect what happens in real-life scenarios. It is feasible that if AUDENZ is widely employed, unwanted outcomes might be discovered that were unseen in the clinical studies.
Mature adults aged 18 to 64 are likely to experience 64% injection site pain, 25% fatigue, 25% headache, 22% malaise, 14% myalgia, 10% arthralgia and 10% nausea, being the most frequent (at least 10%) reported reactions during clinical trials.
In people aged 65 and over, the most common feelings that were reported in studies as a consequence of the injection were pain in the injection site (36%), tiredness (20%), general feeling of discomfort (16%), headache (16%), and joint pain (10%).
In babies and youngsters aged 6 months to 5 years, the most common types of reactions reported in clinical tests (affecting at least 10% of the participants) were tenderness (56%), fussiness (30%), drowsiness (25%), changes in eating behaviors (18%), and fever (16%).
In studies involving kids from ages 6 to 17, the most widespread complaints (affecting over 10% of participants) were pain at the injection site (68%), muscle ache (30%), weariness (27%), ill feeling (25%), headache (22%), lack of appetite (14%), queasiness (13%), and joint ache (13%).
Adults 18 Years Of Age and Older
Information obtained from three trials were studied to evaluate the clinical safety of AUDENZ in adults aged eighteen and above: Trial 1 involved adults aged between 18 and 64 (NCT01776541), Trial 2 looked at adults that were 65 or over (NCT01766921) and Trial 3 assessed the safety of AUDENZ compared to a placebo in adults aged 18 and older (NCT02839330). In all of the studies, participants were given two shots of AUDENZ administered in the muscle 21 days from each other. In each of the three experiments, the responses of both local and systemic adverse reactions were requested for a period of 7 days, while the unsolicited adverse events were monitored for 21 days after each vaccination. For a period of one year after the final vaccination, any significant negative reactions, prespecified reactions indicating possible immune-mediated situations, the emergence of long-term illnesses resulting in a new diagnosis, and incidents necessitating an unplanned medical consultation were monitored. There are 3,579 participants who have been given one or more dosages of AUDENZ within the safety population. There were a total of 3,579 individuals, 1,683 of which were aged 18-64, and the other 1,896 were 65 or older.
This was a US trial consisting of adults over the age of 18, which utilized a randomized, observer-blind, and multicenter control setting. Subjects were split 3:1, with three getting AUDENZ and one getting a saline placebo, and both groups receiving two doses 21 days apart. Altogether, 3,191 examinees aged 18 to 64 (N=1,596) and 65 and above (N=1,595) in the security group got at least one injection of AUDENZ (N=2,395) or dummy drug (N=796). The average age of the people involved in the study who were 18 or older was 58, 45% of them being male. 84% identified as white, 13% as Black/African American, 1% as Asian, fewer than 1% belonging to other races, and 92% not being Hispanic/Latino.
What is the Difference Between Seasonal and H5N1 Avian Influenza?
Both seasonal and bird flu are respiratory infections that share numerous typical early indicators such as a high temperature and achy muscles. Seasonal flu is generally brought about by certain types of A influenza viruses (subtypes H1N1 and H3N2) and type B viruses. It takes place annually, primarily during the late autumn and winter, and is easily transmitted from one person to another. Seasonal flu can be a serious medical condition, particularly for children, pregnant women, and elderly individuals. It is possible for the sickness to necessitate hospital admission and in rare cases, result in death.
Only strains of Type A influenza viruses that fall within the H5N1 subtype are responsible for causing H5N1 avian flu. Avian influenza strain H5N1 tends to proliferate in fowl, yet can be transmitted from birds to humans. This can occur at any time of the year. Infections of H5N1 viruses in humans are exceptionally uncommon, however, when they do happen, it can be life-threatening, leading to fatality in roughly 60% of reported cases. Most people contract H5N1 influenza virus after having direct contact with or being in close proximity to infected birds.
No individuals in the United States have contracted H5N1 influenza.
H5N1 viruses remain in circulation among wild birds, causing the occasional outbreak among domestic poultry; the vast majority of people do not possess any antibodies to the virus. Medical professionals have worries that H5N1 viruses may progress gradually and have the potential to spread easily among individuals.
What Does It Mean that the Vaccine is Adjuvanted?
A substance is added to the vaccine to boost the body’s defense of the vaccinated people, referred to as an adjuvant. With the help of an adjuvant, a tiny amount of influenza protein per immunization is able to bring about the proper protective response in the body and guard against influenza illness. Decreasing the quantity of flu protein per shot contributes to rising the full amount of shots of a dependable and successful vaccine accessible for individuals during a widespread epidemic.
Which Adjuvant is Used in the Vaccine?
The vaccine contains the adjuvant AS03, an oil-in-water emulsion. The AS03 adjuvant consists of D,L-alpha-tocopherol (vitamin E) and squalene, which are oily compounds, as well as polysorbate 80, an emulsifier, in addition to saline water with trace amounts of salt.
The two components of the H5N1 vaccine, H5N1 and AS03 adjuvant, are supplied in two distinct vials. Before it can be utilized, these two vials must be mixed together.
Are there other Adjuvanted Vaccines Approved by FDA for use in the United States?
Yes. This is the first influenza vaccine to be granted FDA approval that has an adjuvant, but it is not the only vaccine to feature this ingredient. For more than 60 years, adjuvants that contain aluminum have been used in vaccinations without much issue. Rarely, they can cause serious local reactions. Cervarix, a vaccine approved by the FDA that protects against cervical cancer, includes AS04 as an adjunct. AS04 is a mixture of aluminum hydroxide and monophosphoryl lipid A. MPL is a purified version of a compound that has been extracted from the membranes of certain bacteria.
This is the first vaccine authorized by the FDA that was designed with the AS03 adjuvant included. This particular adjuvant was used in several flu shots provided to other nations during the swine flu outbreak in 2009.
Is the Vaccine Effective?
The study was conducted across multiple locations to examine the immune system of exactly 2,000 people above eighteen years old. The findings suggested that close to 91% of people aged 18 to 64 and 74% of those aged 65 and over who received the two doses of the vaccine developed antibodies sufficient to reduce the likelihood of contracting H5N1 influenza.
Is the Vaccine Safe?
Yes. A comparison was made between around 3,400 adults over the age of 18 who got the vaccine and around 1,100 who were given the dummy drug, in order to examine the safety. The majority of the clinical study participants reported experiencing discomfort at the injection site. Common side effects included sore muscles, a headache, exhaustion, and inflammation and swelling around the injection site.
Are there any potential risks associated with this vaccine, such as narcolepsy, that have been reported in other nations?
No, that was a different influenza vaccine. The U.S. Food and Drug Administration (FDA) has not authorized the usage of Pandemrix, a vaccine comprised of the AS03 adjuvant and the H1N1 influenza virus subtype, in the United States. GSK is responsible for producing it, but the method used to create Influenza A (H5N1) Virus Monovalent Vaccine, Adjuvanted is not the same as the one used for this.
Pandemrix was used mainly in Europe during the 2009 (H1N1) influenza pandemic in which around 31 million individuals got immunized. The vaccine was not placed into use in the United States.
Narcolepsy is an uncommon but recurring problem with the central nervous system where the brain is not in charge of regulating periods of sleep and being awake. At different points during the day, individuals affected by narcolepsy encounter uncontrollable and abrupt episodes of sleep that can range from a few seconds to several minutes.
A heightened potential for narcolepsy has been observed in children, adolescents, and adults in certain European nations after an injection of Pandemrix. The implications of the increased chance of narcolepsy that was noticed with the Pandemrix vaccine to Influenza A (H5N1) Virus Monovalent Vaccine, Adjuvanted, or to the population in the United States are uncertain since these discoveries may not be relevant to everyone.
Did the FDA take into account the potential danger of developing narcolepsy when they gave the go-ahead to the Influenza A (H5N1) Virus Monovalent Vaccine, Adjuvanted?
Yes. The US Food and Drug Administration evaluated data on the effectiveness and safety of Influenza A (H5N1) Virus Monovalent Vaccine, Adjuvanted before voting to endorse its use. No instances of narcolepsy occurred in the medical examinations of this vaccination. FDA also looked at post-sale security information regarding Pandemrix and the existing epidemiological statistics for affliction caused by H5N1 flu viruses. These viruses have the ability to cause a global epidemic and could produce severe illnesses that would result in a high mortality rate in people who contract them.
The FDA sought out the Vaccines and Related Biological Products Advisory Committee in November 2012 as a means to gain further scientific knowledge on the safety and efficacy of Influenza A (H5N1) Virus Monovalent Vaccine, Adjuvanted. At the meeting, the Committee looked at different matters, including narcolepsy diagnoses in other countries with regards to the Pandemrix vaccine, as well as the research studies which were accessible at the moment. The entire Committee gave a unanimous vote that the evidence of safety and the capability to generate immunity back up the authorization of the Influenza A (H5N1) Virus Monovalent Vaccine, Adjuvanted.
The FDA believes that the pros of the vaccine outweigh the cons due to the huge death rates of those with the H5N1 virus, as well as the shortage of reliable medical interventions which can stop or cure this illness once it has been contracted.
The U.S. Food and Drug Administration will closely watch the security of the Influenza A (H5N1) Virus Monovalent Vaccine, Adjuvanted after it is used in the United States, as is done for all vaccines.
Will the Vaccine be Made Available to the Public?
This vaccine, with an added ingredient, (adjuvanted) to increase its effectiveness, has not been made available for sale commercially. The U.S. The U.S. Department of Health and Human Services has procured the vaccine from Quebec City, Canada’s ID Biomedical Corporation (a subsidiary of GlaxoSmithKline Biologicals) to be included within the U.S. The government has established a collection of resources that can be dispersed by health officials in the event of a crisis.